TEDESCHI Luis
- Ruminant Nutrition, Texas A&M University , College Station, United States of America
- Agricultural sustainability, Animal nutrition modelling, Bioinformatics, Cattle production, Ecology, Farming systems, Grazing management , Gut function, Gut microbiology, Mathematical modelling, Microbial ecology, Microbial fermentation, Physiology, Population dynamics, Precision livestock farming, Rumen microbiology, Rumen microbiome , Ruminant nutrition
- recommender
Recommendations: 3
Reviews: 0
Recommendations: 3
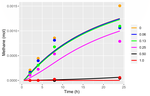
Modelling the impact of the macroalgae Asparagopsis taxiformis on rumen microbial fermentation and methane production
Understanding the mechanisms behind natural bioactive compounds that can potentially reduce methane production in anaerobic conditions. A case study of Asparagopsis taxiformis
Recommended by Luis Tedeschi based on reviews by Alberto Atzori, Henk van Lingen and 2 anonymous reviewersNaturally occurring compounds that can reduce methane production in anaerobic conditions have been studied for quite some time as feasible approaches to mitigate methane production in ruminant animals, especially those of commercial importance. Asparagopsis taxiformis (red algae) and Dictyota bartayresii (brown algae) are effective inhibitors of methane synthesis under in vitro anaerobic fermentation systems (Machado et al., 2014) likely because of their high concentration of secondary metabolites that are toxic to the typical rumen microbiota, including protozoa. In addition to phytoplankton (Palmer and Reason, 2009), Asparagopsis contains a high concentration of haloform compounds (e.g., bromoform, chloroform) while Dictyota has a high concentration of isoprenoid terpenes. Despite the economic and biological impact of diverse phytochemicals on reducing methane production in ruminant animals (Tedeschi et al., 2021), haloform compounds’ environmental impact and safety, in particular, are still unclear. In the present study, Munõz-Tamayo and collaborators (2021) listed relevant literature about the impact of A. taxiformis on ruminal methane production.
Concurrent to the understanding of mechanisms and biology behind the reduction of ruminal methane, mathematical models can lead the way to enhance the effectiveness of feeding A. taxiformis under commercial applications. Thus, in the present study, Munõz-Tamayo and collaborators (2021) sought to develop a mathematical model to understand the rumen fermentation changes in vitro experimentation containing extract of A. taxiformis by adapting a previously documented model by Muñoz-Tamayo et al. (2016).
Modeling development, calibration, and evaluation steps should be independent of each other, requiring complete, distinct, and separate databases (Tedeschi, 2006). However, in rare circumstances where such requirements cannot be met because data availability is scarce, the cross-validation technique, when possible, should be considered to assess data dispersion’s effects on model adequacy. In other situations, clear reasoning for failing to do so must be addressed in the paper. In the present paper, Munõz-Tamayo and collaborators (2021) explained the limitations in their modeling efforts were primarily due to the lack of ideal data: “experiments with simultaneous dynamic data of bromoform, volatile fatty acids, hydrogen, and methane.” Regardless of the availability of ideal data, improvements in the conceptual model are warranted to include amino acids and branched-chain fatty acids fermentation dynamics in the rumen and the fluctuations in ruminal pH.
References
Machado L, Magnusson M, Paul NA, Nys R de, Tomkins N (2014) Effects of Marine and Freshwater Macroalgae on In Vitro Total Gas and Methane Production. PLOS ONE, 9, e85289. https://doi.org/10.1371/journal.pone.0085289
Muñoz-Tamayo R, Chagas JC, Ramin M, Krizsan SJ (2021) Modelling the impact of the macroalgae Asparagopsis taxiformis on rumen microbial fermentation and methane production. bioRxiv, 2020.11.09.374330, ver. 4 peer-reviewed and recommended by PCI Animal Science. https://doi.org/10.1101/2020.11.09.374330
Muñoz-Tamayo R, Giger-Reverdin S, Sauvant D (2016) Mechanistic modelling of in vitro fermentation and methane production by rumen microbiota. Animal Feed Science and Technology, 220, 1–21. https://doi.org/10.1016/j.anifeedsci.2016.07.005
Palmer CJ, Reason CJ (2009) Relationships of surface bromoform concentrations with mixed layer depth and salinity in the tropical oceans. Global Biogeochemical Cycles, 23. https://doi.org/10.1029/2008GB003338
Tedeschi LO (2006) Assessment of the adequacy of mathematical models. Agricultural Systems, 89, 225–247. https://doi.org/10.1016/j.agsy.2005.11.004
Tedeschi LO, Muir JP, Naumann HD, Norris AB, Ramírez-Restrepo CA, Mertens-Talcott SU (2021) Nutritional Aspects of Ecologically Relevant Phytochemicals in Ruminant Production. Frontiers in Veterinary Science, 8. https://doi.org/10.3389/fvets.2021.628445
Effects of feeding treatment on growth rate and performance of primiparous Holstein dairy heifers
Optimizing growth rate of dairy heifers through nutrition to maximize reproduction and production
Recommended by Luis Tedeschi based on reviews by Emilio Mauricio Ungerfeld and 2 anonymous reviewersThe idea of altering the growth rate of replacement heifers to improve reproductive and productive indicators of dairy cattle is not new. In the late 1970s, Gill and Allaire [1] indicated that the first parturition between 22.5 to 23.5 months of age yielded the optimum lifetime performance as long as the heifers had adequate body size [2]. Since 1980s, many studies have been conducted to understand the partitioning of energy between growth and lactation, including the impact of growth rates on the heifer puberty [3] as well as growth and development of the mammary gland [4,5]. The senior author of the recommended study has written previously about this research topic [6].
In the present manuscript, Le Cozler et al. studied the effect of feeding programs to increase the growth rate of late-born heifers to catch up with the growth of those born earlier in the calving season on their reproductive and productive performance. The authors analyzed 217 heifers for three consecutive years, split into three dietary treatments: control (C), accelerated growth rate from birth to 6 months of age (ID1), or accelerated growth rate from birth to 12 months of age (ID2). In this study, the late-born heifers receiving the ID2 treatment were able to partially reach the bodyweight of the early-born heifers at 24 months of age. In part, the incomplete understanding of the prioritization of the use of energy (and other nutrients) for different physiological stages (e.g., maintenance, growth, lactation, and pregnancy) of the dairy animal [7] undercuts the development of more robust feeding strategies to improve the reproductive and productive performance of the animal. In the recommended study by Le Cozler et al., although there was no impact on reproductive performance among groups, heifers in the group ID2 produced less milk (about 400 kg for the whole first lactation) than heifers in the groups C and ID1, apparently suggesting that energy allocation for growth had priority over that needed for lactation. The question then becomes what would have happened with energy partitioning if energy intake was restricted. Studies like this one are important to shed some light on the prioritization of the use of energy and other nutrients in support of growth, pregnancy, and lactation of dairy animals, and how compensatory growth differs between meat versus dairy growing animals, both physiologically and energetically.
References
[1] Gill, G. S., & Allaire, F. R. (1976). Relationship of Age at First Calving, Days Open, Days Dry, and Herdlife to a Profit function for Dairy Cattle1. Journal of Dairy Science, 59(6), 1131–1139. doi: 10.3168/jds.S0022-0302(76)84333-0
[2] Hoffman, P. C. (1997). Optimum body size of Holstein replacement heifers. Journal of Animal Science, 75(3), 836–845. doi: 10.2527/1997.753836x
[3] Cardoso, R. C., Alves, B. R. C., Prezotto, L. D., Thorson, J. F., Tedeschi, L. O., Keisler, D. H., … Williams, G. L. (2014). Use of a stair-step compensatory gain nutritional regimen to program the onset of puberty in beef heifers. Journal of Animal Science, 92(7), 2942–2949. doi: 10.2527/jas.2014-7713
[4] Sejrsen, K., Huber, J. T., Tucker, H. A., & Akers, R. M. (1982). Influence of Nutrition on Mammary Development in Pre- and Postpubertal Heifers1. Journal of Dairy Science, 65(5), 793–800. doi: 10.3168/jds.S0022-0302(82)82268-6
[5] Sejrsen, K., & Purup, S. (1997). Influence of prepubertal feeding level on milk yield potential of dairy heifers: a review. Journal of Animal Science, 75(3), 828–835. doi: 10.2527/1997.753828x
[6] Le Cozler, Y. L., Lollivier, V., Lacasse, P., & Disenhaus, C. (2008). Rearing strategy and optimizing first-calving targets in dairy heifers: a review. Animal, 2(9), 1393–1404. doi: 10.1017/S1751731108002498
[7] Tedeschi, L. O., and D. G. Fox. 2018. The Ruminant Nutrition System: An Applied Model for Predicting Nutrient Requirements and Feed Utilization in Ruminants. (2nd ed.). XanEdu, Acton, MA."

Lactation curve model with explicit representation of perturbations as a phenotyping tool for dairy livestock precision farming.
Developing smart fitting algorithms to identify random perturbations in time-series data
Recommended by Luis Tedeschi based on reviews by Alberto Atzori, Jennifer Spencer and 1 anonymous reviewerThe ability to adequately characterize the lactation curve of livestock is important not only to ensure proper nutrition of the lactating animal but, among many other benefits, it can assist in diagnosing the incidence of diseases, predicting the quantity of milk production, and comparing animals within the herd for managerial strategies such as culling. Eventually, such smart fitting algorithms can lead to improved genetic selection of more productive animals after genetic-unrelated noises are removed from the data, systematically.
The manuscript by Ben Abdelkrim et al. developed and explained an algorithm to detect perturbations in lactation curves of dairy goats. Researchers have been interested in accurately describing lactation curves since the early-1960s. Johansson [1] proposed a nonlinear decay function, Nelder [2] described an inverse polynomial, and Wood [3] proposed the incomplete gamma function to describe milk production of dairy cows. Unfortunately, many of the lactating animals that yielded lactation curves that did not comply with the typical, expected pattern of milk production were usually discarded and, until then, efforts to address this lack of adherence were not conducted. The recommended manuscript provides a different perspective in which rather than discarding the lactation profile, one can model the perturbations of the lactation curve as an attempt to identify possible problems (e.g., mastitis) and minimize their occurrence. Such an algorithm is important to separate females that show resilient attributes from those females that show sustainable attributes, as per existing definitions proposed by Tedeschi et al. [4].
The recommended manuscript proposes the Perturbed Lactation Model to explicitly account for multiple perturbations in the time-series milk production in dairy goats. When perturbations occur in biological processes, a typical negative impact is observed in the animal’s response, but on rare occasions, positive impacts can occur. In this case, the animal responds positively to the perturbation (i.e., responsive), and as a result, there is an increase in their output when compared to unperturbed animals. The recommended manuscript only considered negative impacts due to perturbations in the lactation curve of dairy goats. Future modifications should include positive responses due to perturbations. In this case, animals would be “positively responsive” to perturbations, and examples of such behavior include feed intake and growth curves.
References
[1] Johansson, I. (1961). Genetic Aspects of Dairy Cattle Breeding. University of Illinois Press, Urbana, IL.
[2] Nelder, J. A. (1966). Inverse polynomials, a useful group of multi-factor response functions. Biometrics. 22 (1):128-141. doi: 10.2307/2528220
[3] Wood, P. D. P. (1967). Algebraic model of the lactation curve in cattle. Nature. 216 (5111):164-165. doi: 10.1038/216164a0
[4] Tedeschi, L. O., J. P. Muir, D. G. Riley, and D. G. Fox. (2015). The role of ruminant animals in sustainable livestock intensification programs. Int. J. Sustainable Dev. World Ecol. 22 (5):452-465. doi: 10.1080/13504509.2015.1075441